Product Development
New Drug Development Process
New Drug Development
Tou Pharma Bio Pvt. Ltd develops formulations for new chemical entities, OTC products, generic products, and 505(b)(2) product candidates. The company has also evolved the following dosage forms.
- 1.Tablets
- 2.Capsules, powder-filled
- 3.Capsules, liquid-fi...

New Drug Development and Strategic Planning
Tou Pharma Bioscientists along with regulatory agencies have worked efficiently and effectively move a drug candidate forward. The major activities include the following.
- 1.Regulatory strategic planning
- 2.CMC support and preparation
- 3.FDA Pre-IND support and preparation
- 4.FDA meetings ...

Formulation Development Services
To optimize the formulation and scale-up for the cGMP manufacturing of clinical trial materials for Phase I and Phase II trials, Tou Pharma Bio’ formulation development team and manufacturing unit worked together. Tou Pharma Bio cGMP manufacturing team is under the jurisdiction of the Quality Assurance Unit and complies with Tou Phar...
Liquid Dosage Form and Capsule Platform Technologies
Tou Pharma Bio is a pioneer in the development of liquid-filled hard gelatin and HPMC capsule formulation technologies. The physical and chemical characteristics of a drug compound are determined by the related technologies. These characteristics may include the following.
- Sensitivity to water
- Poor solubi...

Analytical & Bioanalytical Service
Analytical​ Bioanalytical Chemistry
Tou Pharma Bio’s experienced team of analytical chemists in the field of development and validation of stability developed methods to be used to quantitate drug substance purity and percentage drug content in a dosage form, including impurities and degradation prod...
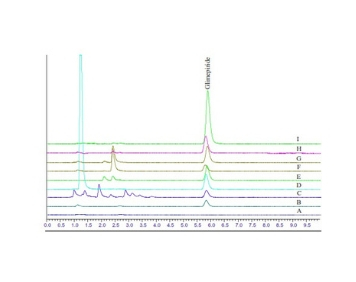
Formal stability studies
Formal stability studies
Phase I / Phase II Formal Stability Studies
As per the ICH Guidelines Q1A and Q1B, Tou Pharma Bio have provided the stability protocol design and testing for bulk drug substances and drug products. The stability data has been generated by our Quality Contr...

Packaging Material DMF & Excipients - Type III & IV
Packaging Material DMF & Excipients - Type III & IV
Primary Packaging material is the material which is directly is contact with the Product and the same material holds the product. The main purpose of Primary packaging is to protect the product from any chemical, biological, climatic or any other hazards which...

